What is Sonanos™?
Sonanos™:
Hyaluronic acid nanogel
A Next-Generation Pharmaceutical Excipient
for Injectable Formulations to Prolong Drug
Release and Enhance API Solubility

Sonanos™ (hyaluronic acid nanogel) is a cutting-edge pharmaceutical excipient primarily intended for injectable formulations that is currently awaiting first-in-human trials. Sonanos™ can act as an API carrier, enabling its use as a sustaned release agent for biologics or as a solubilizer for higher-molecular-weight modalities.
What Is Hyaluronic Acid Nanogel?
Hyaluronic acid nanogels are nanoscale, hyaluronic acid–based hydrogels that are typically spherical in shape and range from 1 to 1,000 nanometers in diameter. Owing to their three-dimensional network structure, these nanogels can encapsulate a wide variety of active pharmaceutical ingredients (APIs) within the gel matrix, serving as promising therapeutic carriers in biomedical applications.
Although most hyaluronic acid nanogels remain at the academic research stage, Asahi Kasei Corporation is actively pursuing their commercialization as a novel pharmaceutical excipient to provide pharmaceutical companies with an innovative approach to overcoming formulation challenges.
Reference:
S. S. Myint et al. Pharmaceutics 2023, 15, 2671.
https://doi.org/10.3390/pharmaceutics15122671
Formulation Challenges We Address: Limited Options to Modulate Release Profile & Poor Solubility of High MW Compounds
Drug discovery is increasingly shifting toward higher‑molecular‑weight modalities such as proteins, including CD‑targeted bispecific antibodies, and targeted protein degraders (TPDs), leading to formulation challenges like limited options to modulate release profiles and poor solubility.
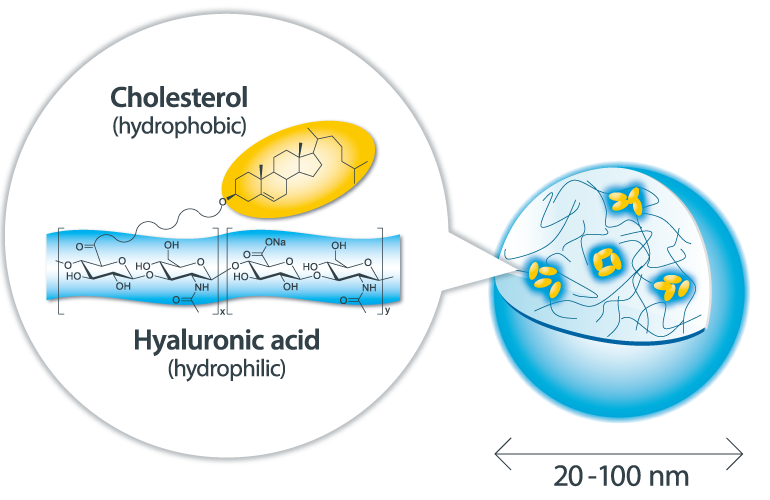
Sonanos™: Cutting-Edge API Carrier to Prolong Drug Release and Enhance API Solubility
These challenges and demands in formulation research have motivated us to investigate a novel pharmaceutical excipient, Sonanos™ (hyaluronic acid nanogel). The main component of Sonanos™ is a nano-sized hydrogel particle (nanogel) capable of encapsulating a wide range of APIs—including proteins (such as CD-targeted bispecific antibodies), peptides, and low‑molecular‑weight compounds— through simple mixing in aqueous solution, without causing denaturation. Sonanos™ enables subcutaneous sustained release of APIs via in-situ depot formation and significantly enhances the solubility of poorly water-soluble APIs.

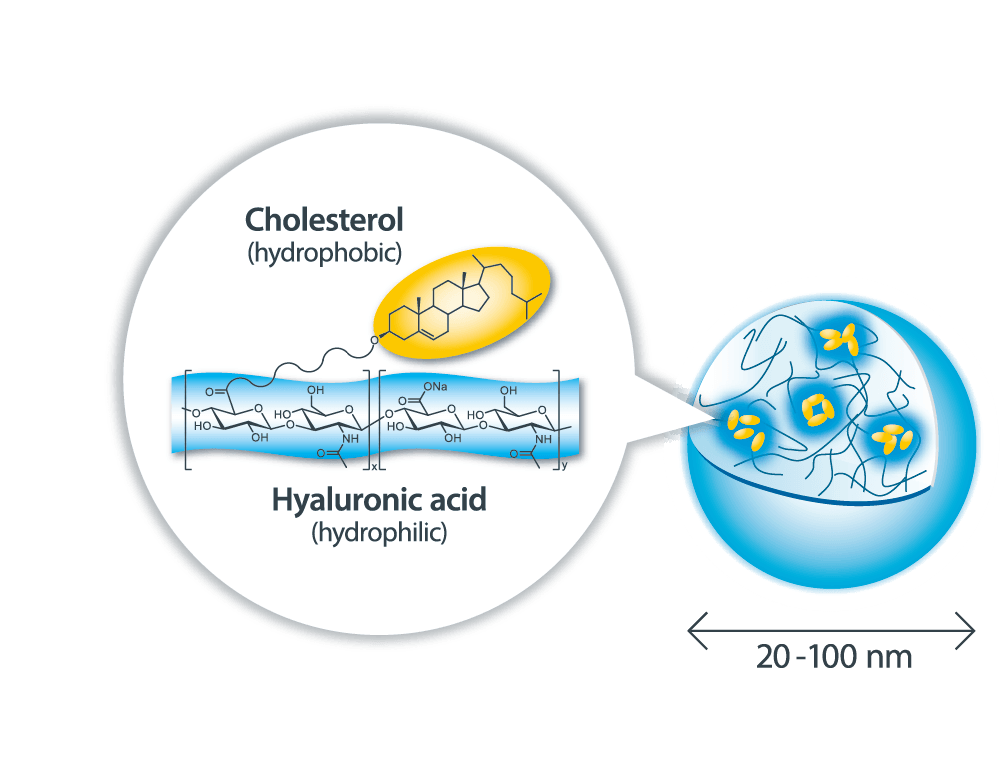
Why Can Sonanos™ Encapsulate APIs?
The main component of Sonanos™ is a nano‑sized hydrogel particle (nanogel) composed of Cholesterol‑Modified Hyaluronic Acid (CHHA). APIs are encapsulated primarily through hydrophobic interactions, whereby the API becomes entrapped within the cholesterol domains inside the nanogel.
Grade Lineup
We offer two grades. Sustained Release Grade (Sonanos™ PG) forms a depot upon exposure to physiological salt, enabling in situ subcutaneous sustained release. Solubility Enhancement Grade (Sonanos™ DS), with a higher degree of cholesterol modification, improves the solubility of poorly water‑soluble APIs and is suitable for both subcutaneous and intravenous administration.

Formulation Process of Sonanos™
Appearance & Composition of Sonanos™
We deliver Sonanos™ as a cotton-like, freeze-dried solid composed of nanogel (assembly of cholesterol-modified hyaluronic acid, CHHA) and sucrose, which can be reconstituted into a nanogel dispersion by adding water.

Typical Formulation Process for Sonanos™: Very Simple
Formulating with Sonanos™ is straightforward: the nanogel encapsulates APIs through simple mixing. First, solubilize Sonanos™ in water. Once the aqueous Sonanos™ solution is prepared, add the API and stir to encapsulate it into the particles. You may also use an isotonic agent, such as sucrose. After the formulation is obtained, sterilization can be achieved using a 0.22µm filter.

Requirements for Formulation Production Facility
No need for special facilities
Encapsulation of API into Sonanos™ can be achieved by a simple mixing process and aseptic filtration.
*Optimization of formulation process parameters may be required to improve the loading efficiency.
Recommendations for CMO (if used)
Sonanos™ does not restrict the choice of CMO, unless the Sonanos™ concentration is too high to conduct sterile filtration.
Typical Formulation Composition
Below is the typical formulation composition with a dosing volume of 1 mL when using Sonanos™. The envisioned API dose would be approximately 2.5-14mg for a 1mL dosing volume, which is the typical maximum dosing volume for subcutaneous (SC) injection.
Sonanos™ PG
| Component | Weight | Weight ratio | Conc. |
|---|---|---|---|
| API | 2.5-10 mg | 0.1-0.4 wt | 2.5-10 mg/mL |
| CHHA | 25 mg | 1.0 wt | 25 mg/mL |
| Phosphate buffer | 0-1.3 mg | 0-0.05 wt | 0-10 mM |
| Isotonic agent | 100 mg | 4.0 wt | 100 mg/mL |
| Water | 1000 mg | – | – |
Sonanos™ DS
| Component | Weight | Weight ratio | Conc. |
|---|---|---|---|
| API | 3.5-14 mg | 0.1-0.4 wt | 3.5-14 mg/mL |
| CHHA | 35 mg | 1.0 wt | 35 mg/mL |
| Phosphate buffer | 0-1.3 mg | 0-0.04 wt | 0-10 mM |
| Isotonic agent | 100 mg | 2.8 wt | 100 mg/mL |
| Water | 1000 mg | – | – |
Benefits to customers (pharmaceutical companies)
Multifunctional
Tunable solubility enhancement and sustained release profiles through grade selection.
API versatility
Capable of loading a wide range of small molecules, peptides, and proteins with various molecular attributes.
Convenience
Formulation can be done simply and easily just by adding Sonanos™.
Safety
Sonanos™ is derived from naturally occuring substances that have high biocompatibility.
Benefits to end-users (patients)

Reduced patient
burden
Fewer injections are needed, reducing the burden on patients.
Reduced side effects
Sonanos™ is made of materials with low toxicity, which reduces concerns about side effects derived from excipients.
Improvement in
anticipated effects
Solving formulation issues, such as poor water solubility, can increase expectations for the anticipated effects of new pharmaceuticals development.
Improvement in
quality of life
Reducing the burden, anxiety and side effects on patients, and raising expectations for the development of new pharmaceuticals, will contribute to the overall improvement of patients’ quality of life.
