Sustained Release Agent
Application of Sonanos™:
Sustained Release Agent for Biologics
Sonanos™ PG (Sustained Release Grade) forms an in situ depot triggered by physiological salt, providing a platform for sustained API release. It enables suppressed burst release and prolonged release duration for a broad range of APIs, including proteins, long‑acting peptides, and poorly water‑soluble peptides.
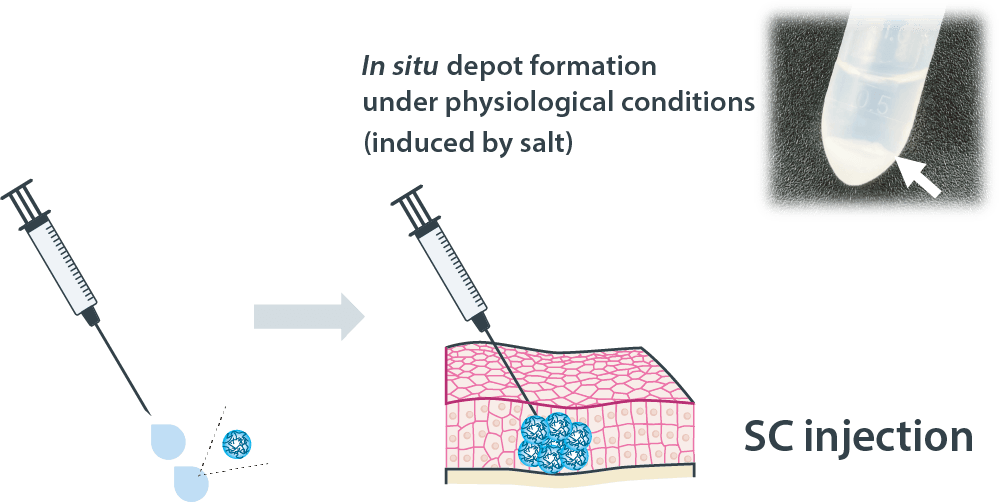
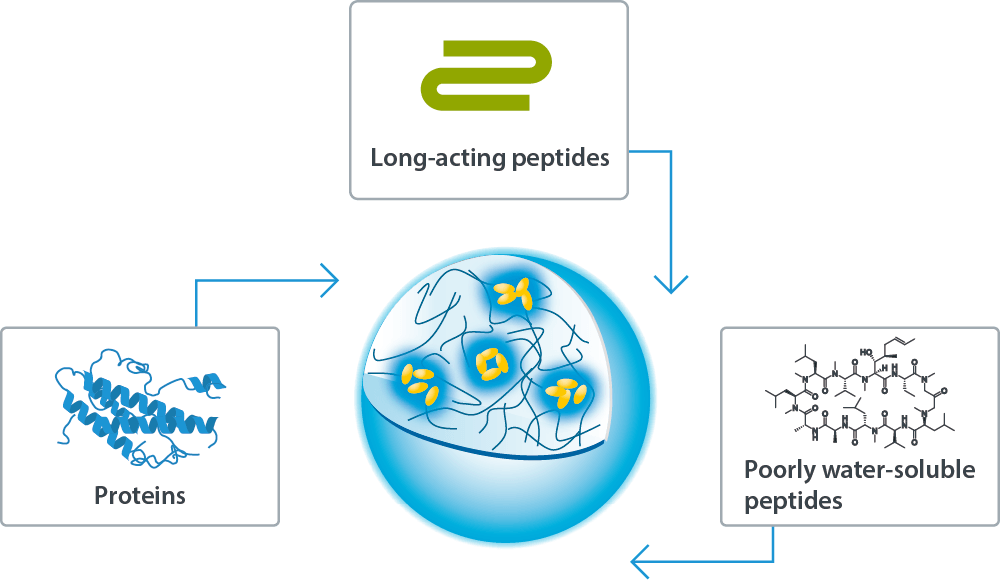
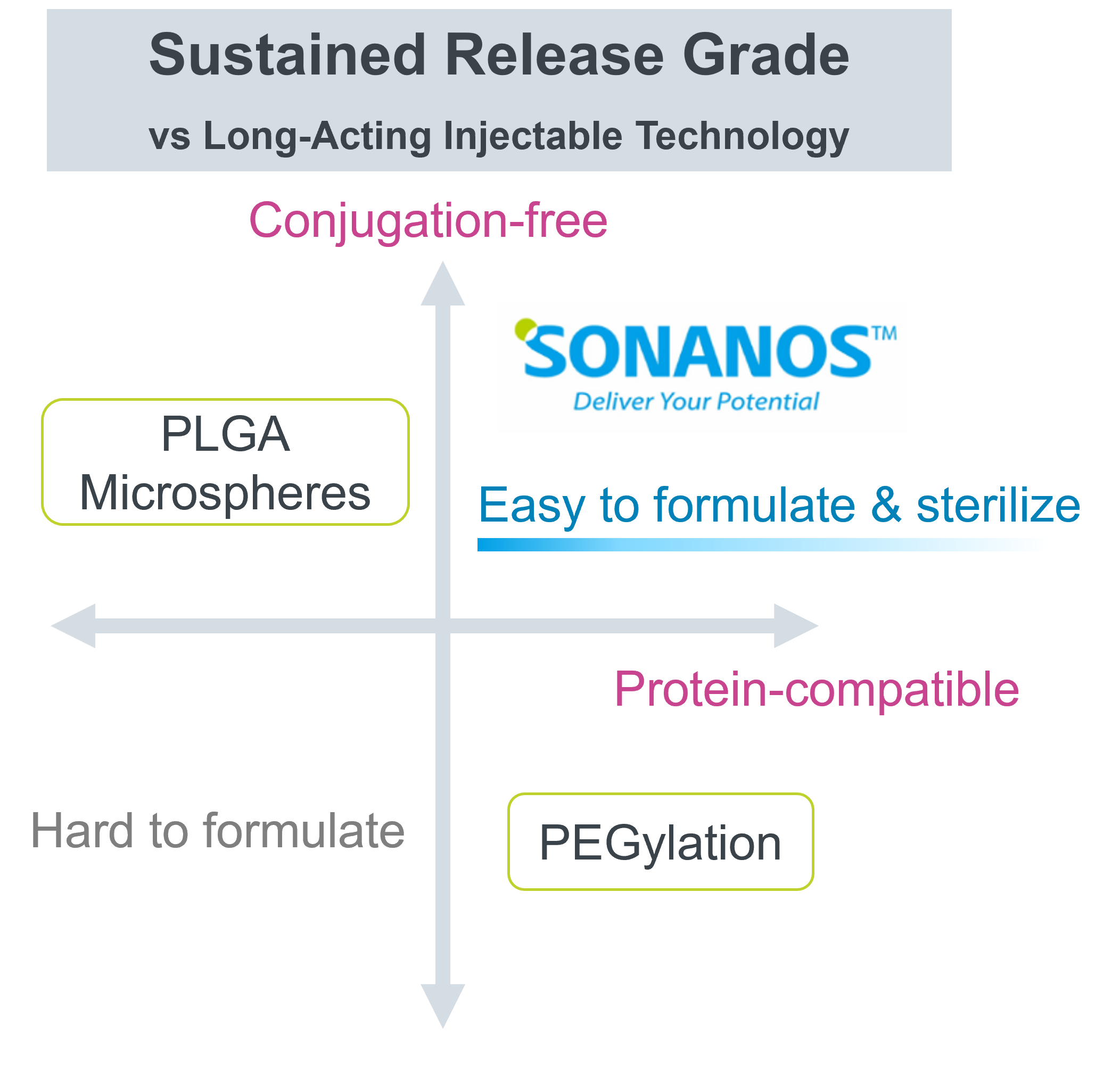
Compared with conventional sustained‑release technologies (e.g., PLGA microspheres) and half‑life‑extension technologies (e.g., PEGylation), Sonanos™ PG offers three key advantages:
- It does not use organic solvents during encapsulation of API—making it suitable even for proteins that easily lose activity in such solvents.
- It does not alter the chemical structure of API.
- It is easy to formulate with Sonanos™—requiring only a simple mixing process—and it can be sterilized by filtration.
Typical Formulation Composition
Shown below is a representative formulation using Sonanos™ PG with a dosing volume of 1 mL. The envisioned API dose ranges from approximately 2.5 to 10 mg per 1 mL, which corresponds to the typical maximum dosing volume for subcutaneous (SC) injection.
Sonanos™ PG
| Component | Weight | Weight ratio | Conc. |
|---|---|---|---|
| API | 2.5-10 mg | 0.1-0.4 wt | 2.5-10 mg/mL |
| CHHA | 25 mg | 1.0 wt | 25 mg/mL |
| Phosphate buffer | 0-1.3 mg | 0-0.05 wt | 0-10 mM |
| Isotonic agent | 100 mg | 4.0 wt | 100 mg/mL |
| Water | 1000 mg | – | – |
Sustained Release of Proteins
A key advantage of Sonanos™ is its compatibility with proteins, enabling sustained release without protein
denaturation due to the absence of organic solvents during formulation.
As an example, human growth hormone (hGH) was used as a model protein. A formulation prepared by simple mixing of
Sonanos™ and hGH was administered subcutaneously to Sprague–Dawley rats (SD rats). Plasma pharmacokinetic (PK)
analysis demonstrated that hGH was continuously released over approximately one week.
Plasma PK of human growth hormone (hGH) after SC in rats
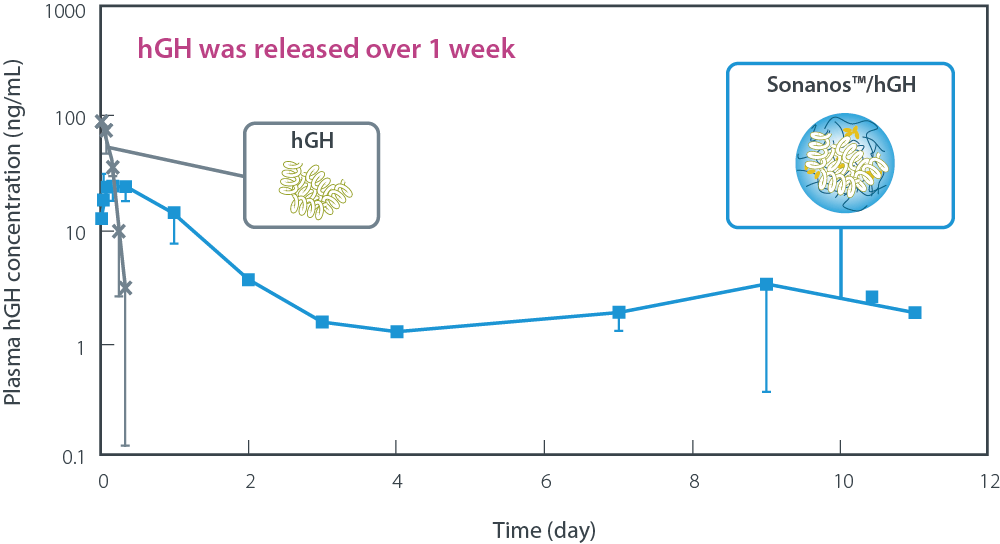
- SD Rat (CRJ)、♂、6w
- N=3
- SC of 5 mL/kg
- Detected by ELISA
Evaluation of In Vitro Bioactivity
To assess whether encapsulation affects protein bioactivity, we conducted a cell viability assay comparing
Sonanos™ PG‑encapsulated protein with the native form.
TNF‑α was used as a model protein.
The assay was performed using L929 cells treated with TNF‑α in the presence of actinomycin D,
hydroxypropyl‑β‑cyclodextrin (HP‑β‑CD), and sucrose. HP‑β‑CD served as a nanogel disintegrator to facilitate TNF‑α
release from the nanogel particles.
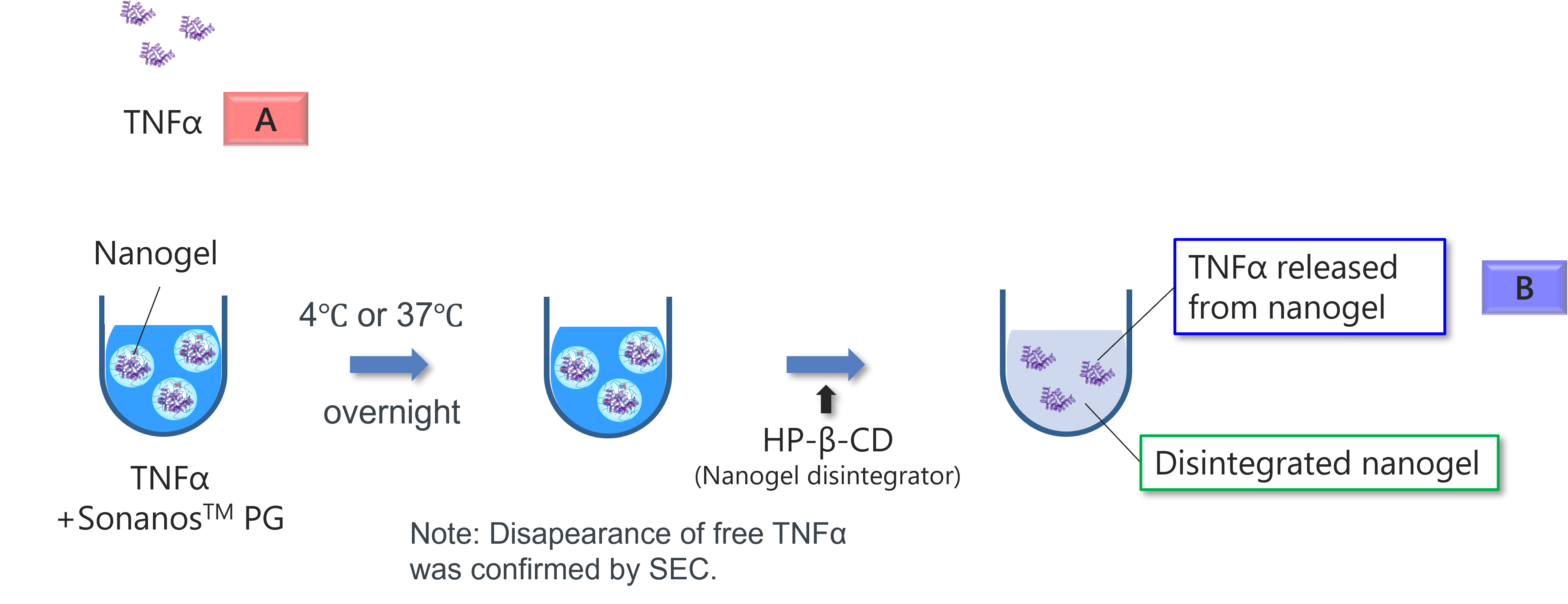
Encapsulation in Sonanos™ PG did not compromise TNF‑α bioactivity. The EC₅₀ values for native TNF‑α and for Sonanos™ PG‑encapsulated TNF‑α following incubation at both 4 °C and 37 °C showed no significant differences, indicating preserved biological activity after encapsulation.

Note: The slight increase in bioactivity of TNFα observed upon encapsulation in Sonanos™ PG is due to the prevention of TNFα adsorption to the plate by Sonanos™ PG. However, this effect would not affect the conclusion.
Sustained Release of Long-acting peptides
Sonanos™ PG is also effective for extending the release duration of
long‑acting peptide APIs, a rapidly growing
modality in pharmaceutical development.
In one study, semaglutide was used as a model drug.
A formulation consisting of semaglutide mixed with Sonanos™ PG
was subcutaneously administered to Sprague–Dawley rats, and PK profiles were evaluated.
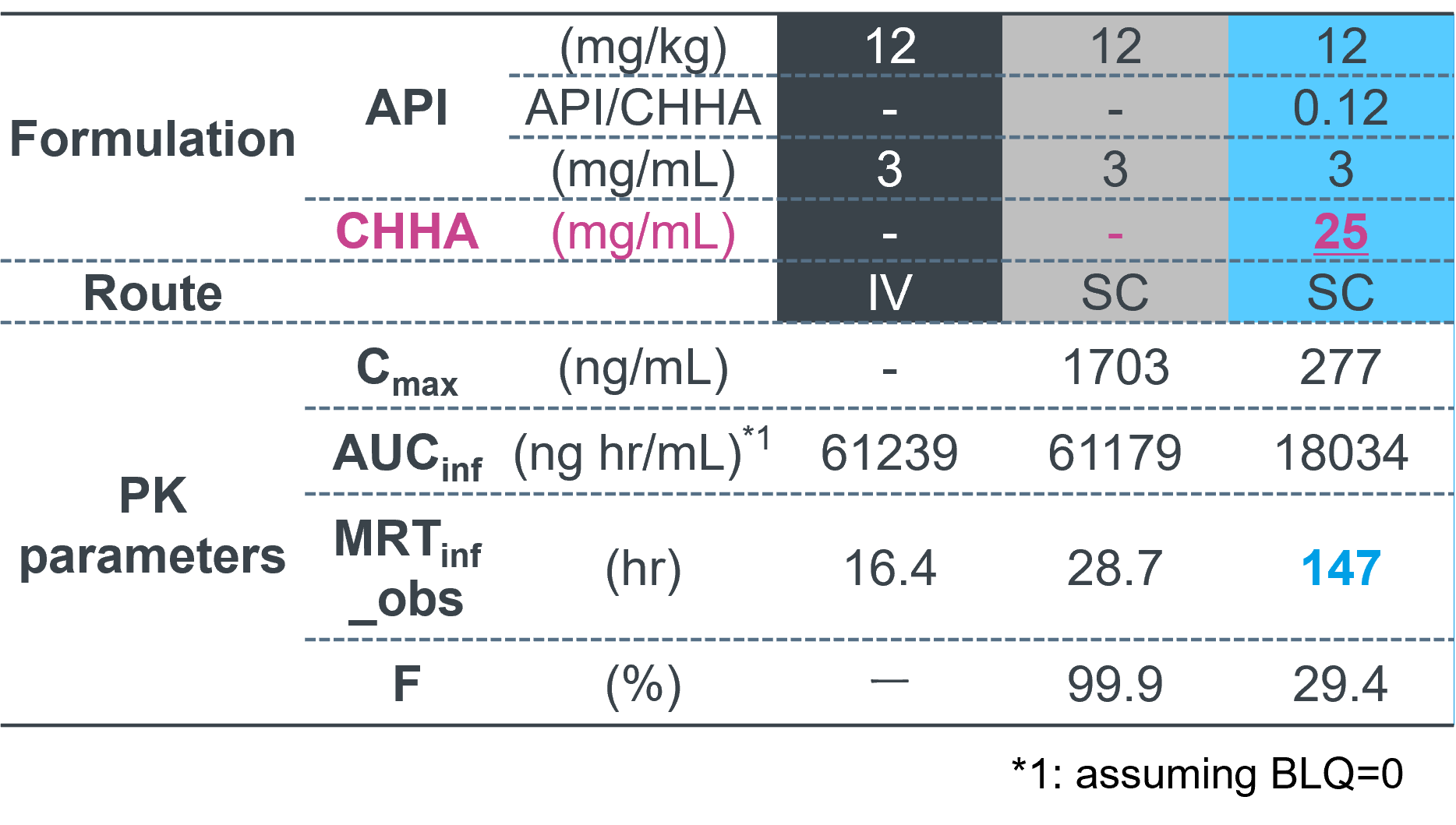
Compared with formulations containing the same dose of semaglutide alone,
the Sonanos™ PG formulation showed
marked suppression of burst release
and a significantly prolonged release period.
For example, when the target therapeutic window was defined as 1,000–100,000 ng/mL,
sustained exposure was maintained for approximately
10 days at a dose of 40 mg/kg.
Plasma PK of semaglutide after SC in rats
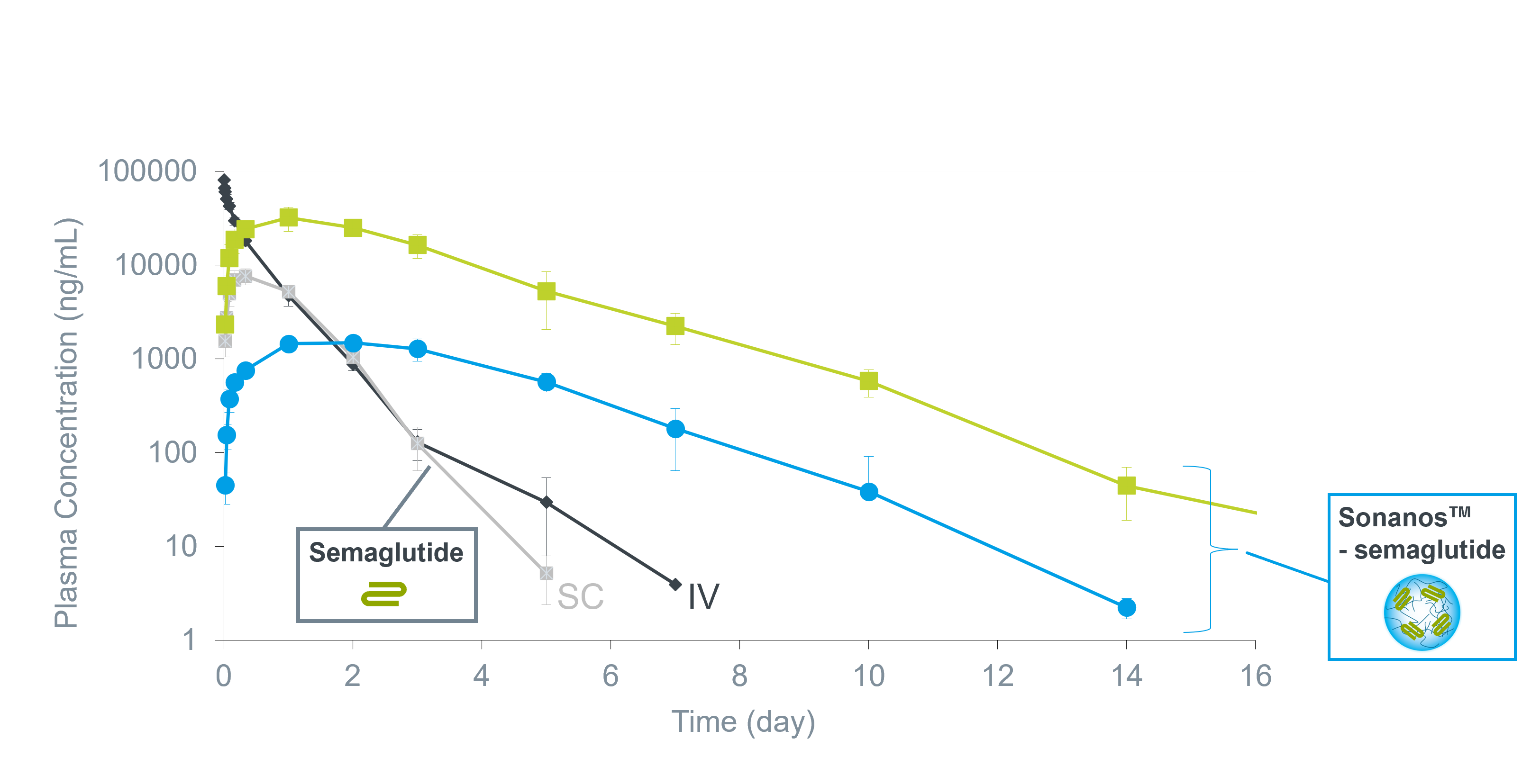
- SD Rat (CRJ)、♂、6-8 w
- N=3
- SC of 4 mL/kg
- Detected by LC/MS/MS
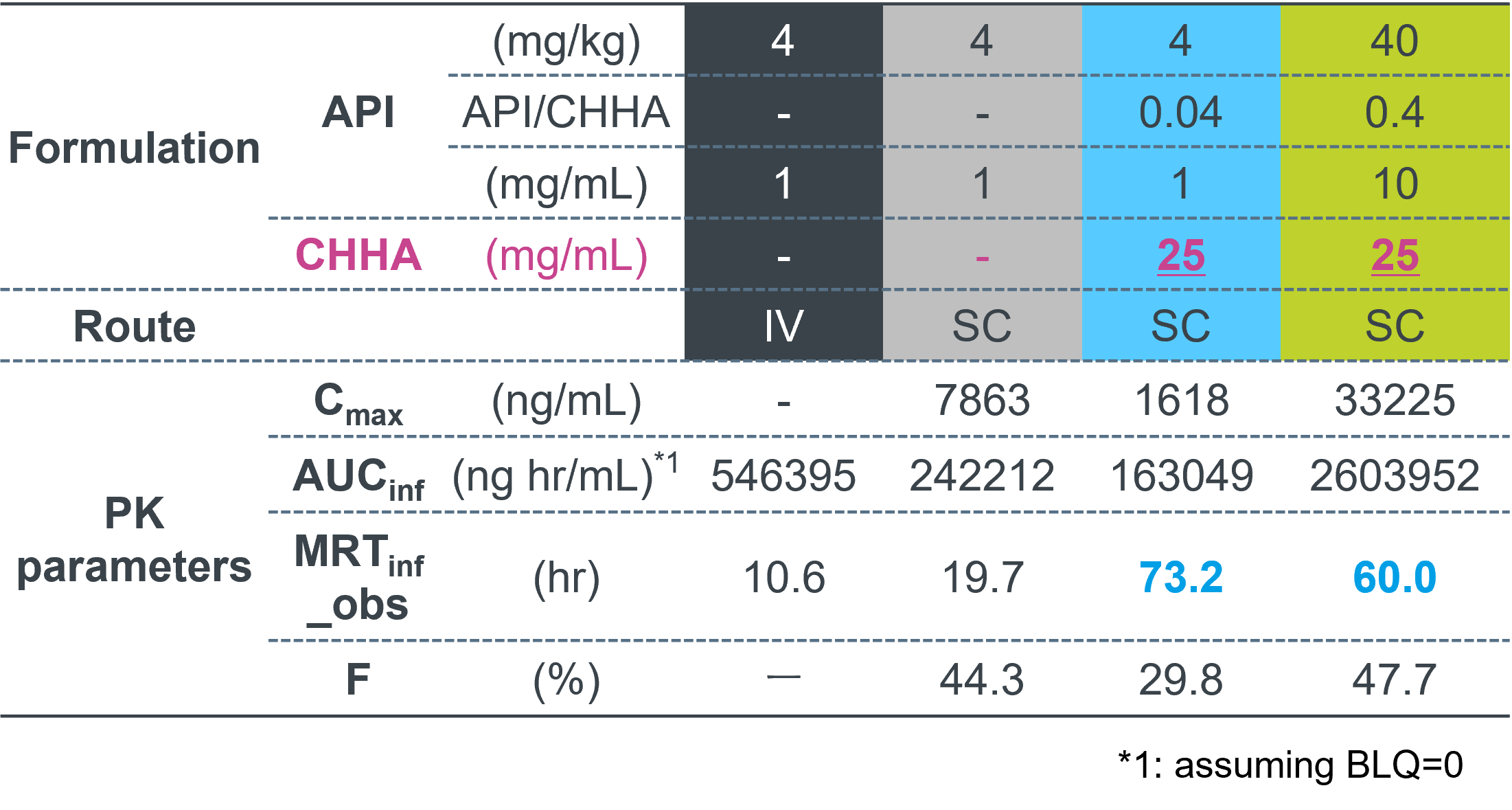
Formulation compositions & PK parameters
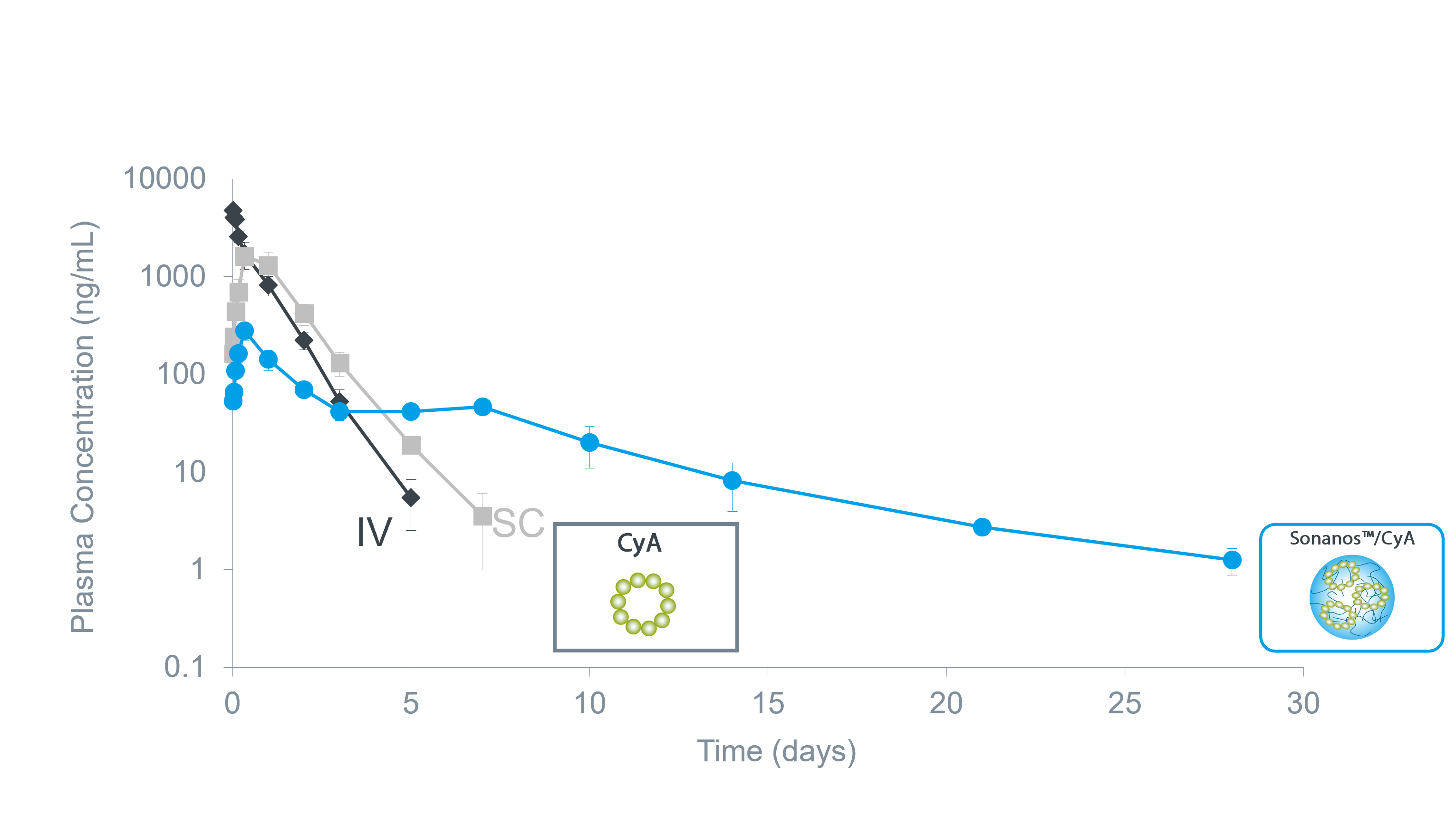
Sustained Release of Poorly water-soluble peptides
Sonanos™ PG can also function as a sustained‑release platform for
poorly water‑soluble peptide APIs.
In this study,
cyclosporin A (CyA) was used as a model compound.
Notably, formulation with Sonanos™ PG achieved a
prolonged release duration of up to approximately 28 days.
This extended release is attributed to the strong interaction between the API and the hyaluronic acid nanogel,
which further stabilizes the depot structure.
Plasma PK of cyclosporin A (CyA) after SC in rats
- SD Rat (CRJ)、♂、6-8 w
- N=4
- SC of 4.0 mL/kg
- Detected by LC/MS/MS
Formulation compositions & PK parameters