Solubilizer
Application of Sonanos™:
Solubilizer for Higher-Molecular-Weight Modalities
Sonanos™ DS (Solubility Enhancement Grade) dramatically enhances the solubility of poorly water‑soluble APIs, including higher-molecular-weight modalities such as peptides. Simple addition of the API powder to a Sonanos™ DS solution enables effective solubilization without the need for organic solvents.

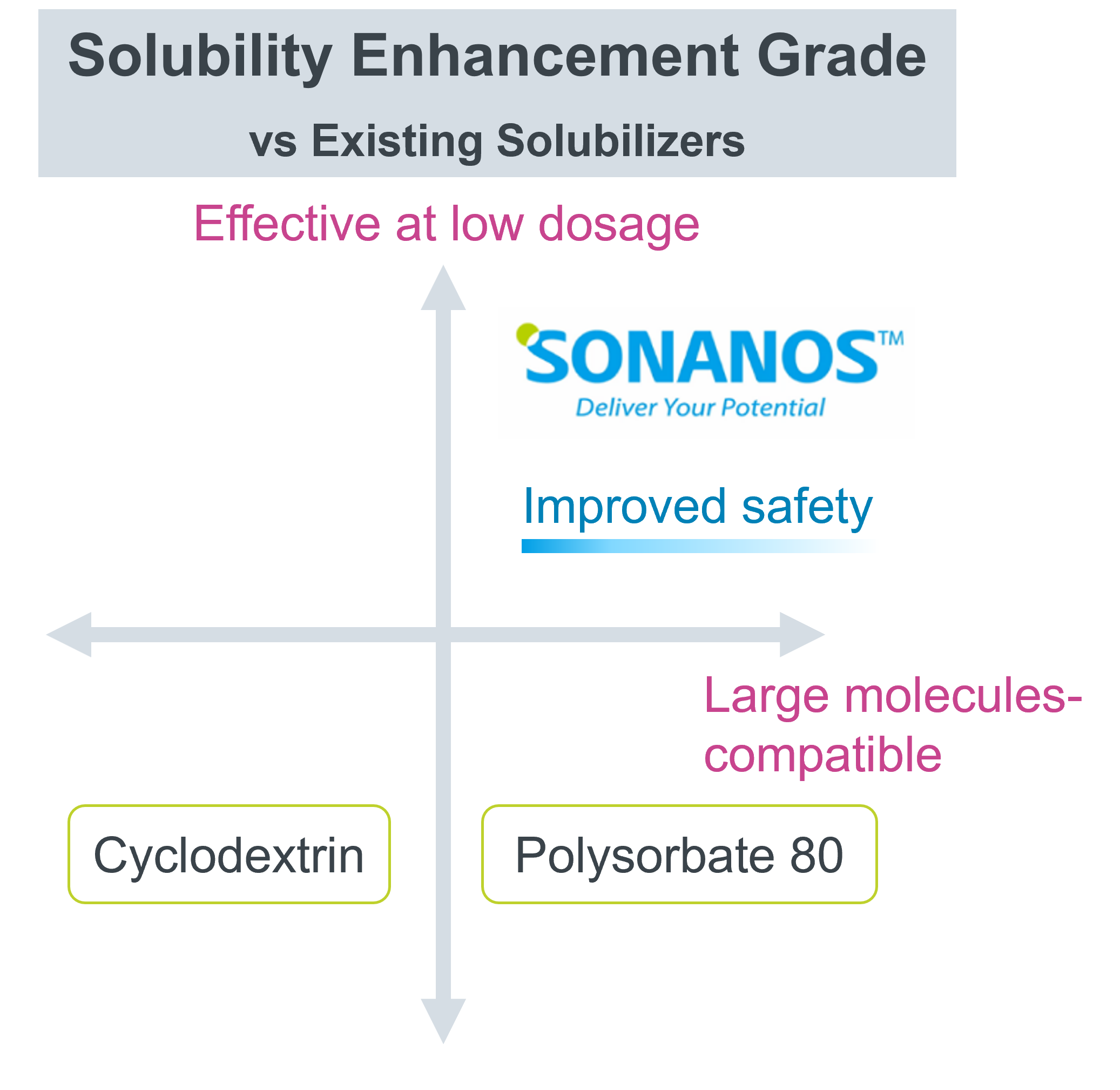
Compared with conventional solubilizers, Sonanos™ DS offers three advantages:
- It can achieve solubilization with a smaller amount of excipient.
- It is applicable even to components that have traditionally been difficult to solubilize —such as large molecules with a molecular weight over 800 (e.g., poorly water‑soluble peptides or TPDs).
- It demonstrates a high level of safety.
Typical Formulation Composition
Below is a representative formulation using Sonanos™ DS with a dosing volume of 1 mL. The envisioned API dose ranges from approximately 3.5 to 14 mg per 1 mL,, which corresponds to the typical maximum dosing volume for subcutaneous (SC) injection.
Sonanos™ DS
| Component | Weight | Weight ratio | Conc. |
|---|---|---|---|
| API | 3.5-14 mg | 0.1-0.4 wt | 3.5-14 mg/mL |
| CHHA | 35 mg | 1.0 wt | 35 mg/mL |
| Phosphate buffer | 0-1.3 mg | 0-0.04 wt | 0-10 mM |
| Isotonic agent | 100 mg | 2.8 wt | 100 mg/mL |
| Water | 1000 mg | – | – |
Solubility Enhancement of Poorly Water-Soluble Peptides
Sonanos™ DS is an effective solubilizer for poorly water‑soluble peptides. For example,
we evaluated its solubility‑enhancing effect using poorly water‑soluble cyclic peptide powders.
Sonanos™ significantly improved peptide solubility without the use of organic solvents.
Although specific molecular structures are confidential,
in one case (cyclic peptide A), a commercially available solubilizer (polysorbate 80) increased solubility by
approximately
2,000‑fold compared with the API alone, whereas
Sonanos™ DS (2.4%) achieved an improvement exceeding 70,000‑fold.
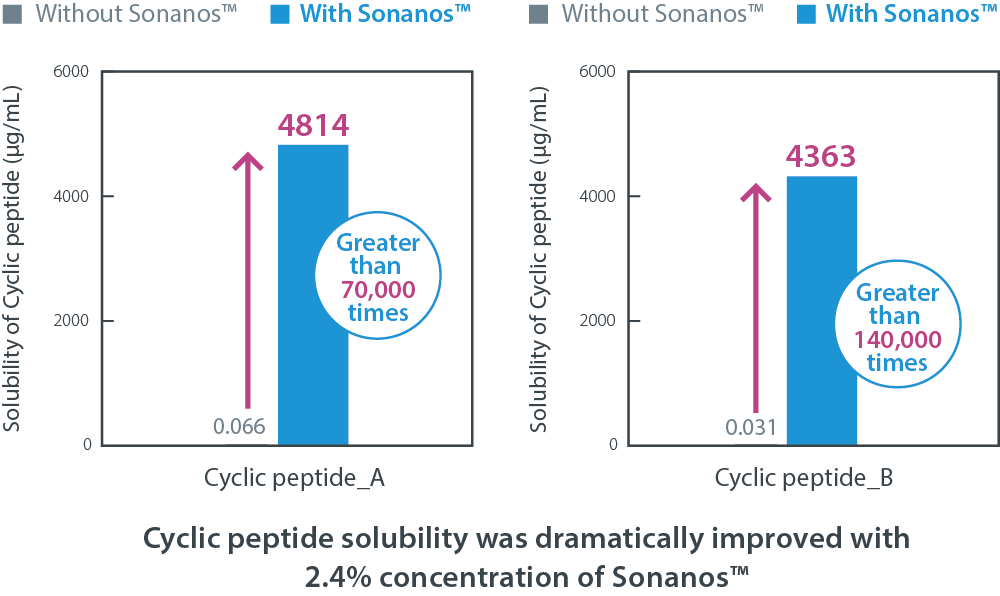
Note: moleular weight of cyclic peptide A and B is 2,201 and 1,036, respectively.
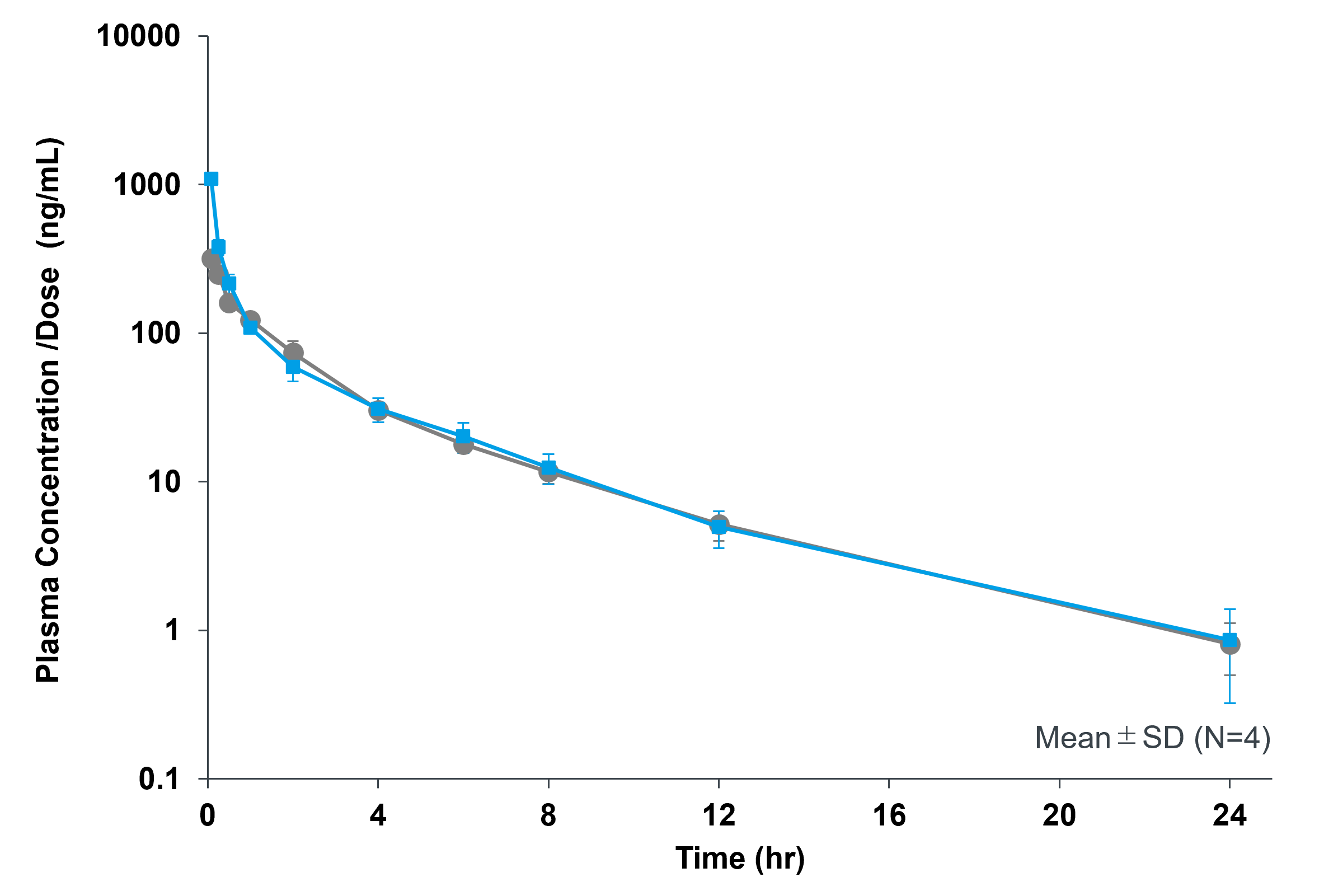
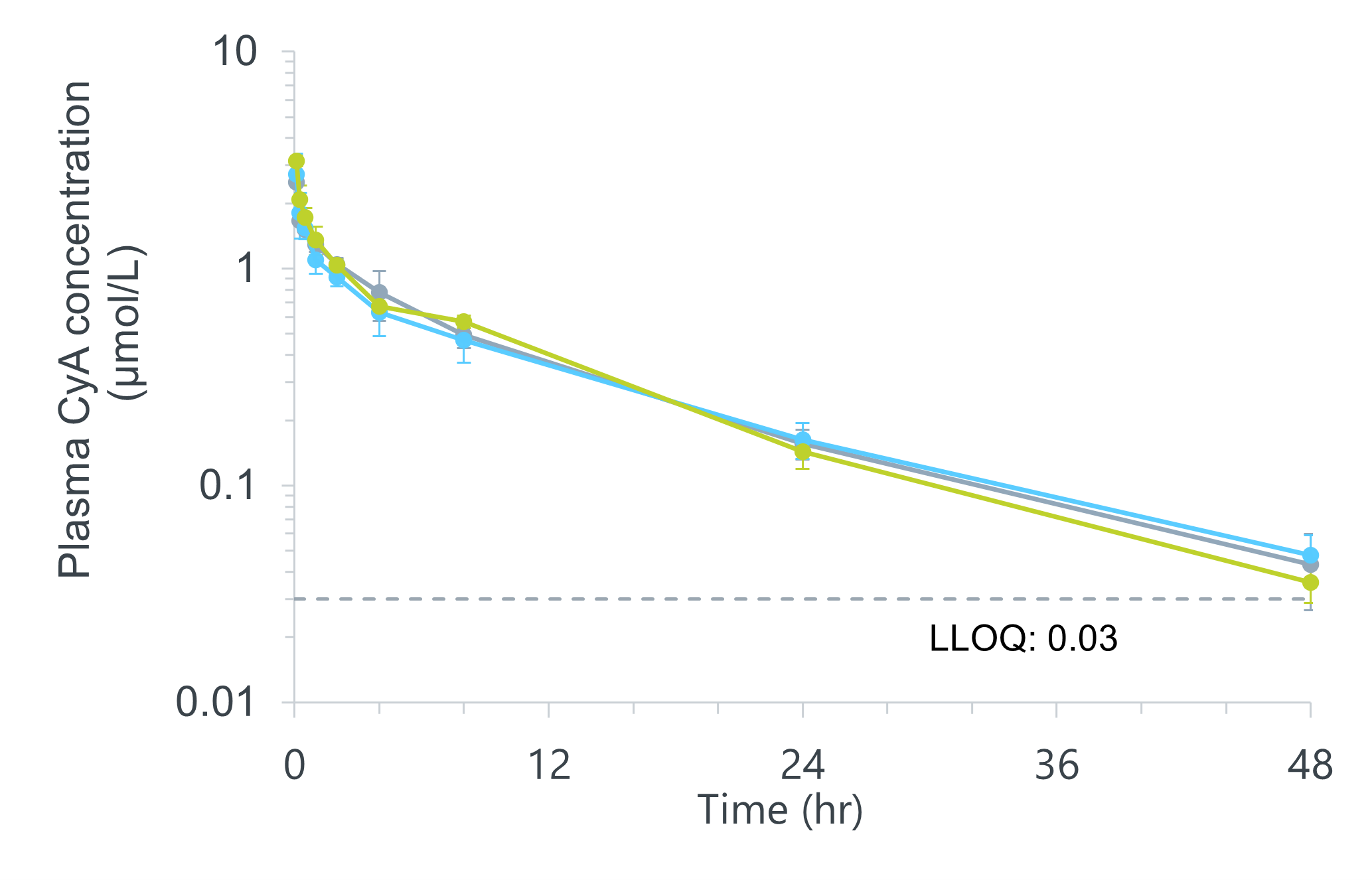
Effect of Sonanos™ DS on PK Profile of Cyclic Peptides
We also investigated the effect of Sonanos™ DS on the pharmacokinetic (PK) profile of a poorly water‑soluble
peptide.
Cyclosporin A (CyA) was used as a model drug,
solubilized with Sonanos™ DS, and intravenously administered to Sprague–Dawley rats (SD rats).
Sonanos™ DS did not significantly alter the PK profile of CyA, indicating its suitability as a solubilizer.
These results suggest that APIs encapsulated in Sonanos™ nanogel particles are rapidly released after entering
systemic circulation.
- Animals: SD rats (Slc:SD), male, 6-7 weeks
- IV, N = 3, 2.5 mL/kg
- Concentration measurements: UHPLC

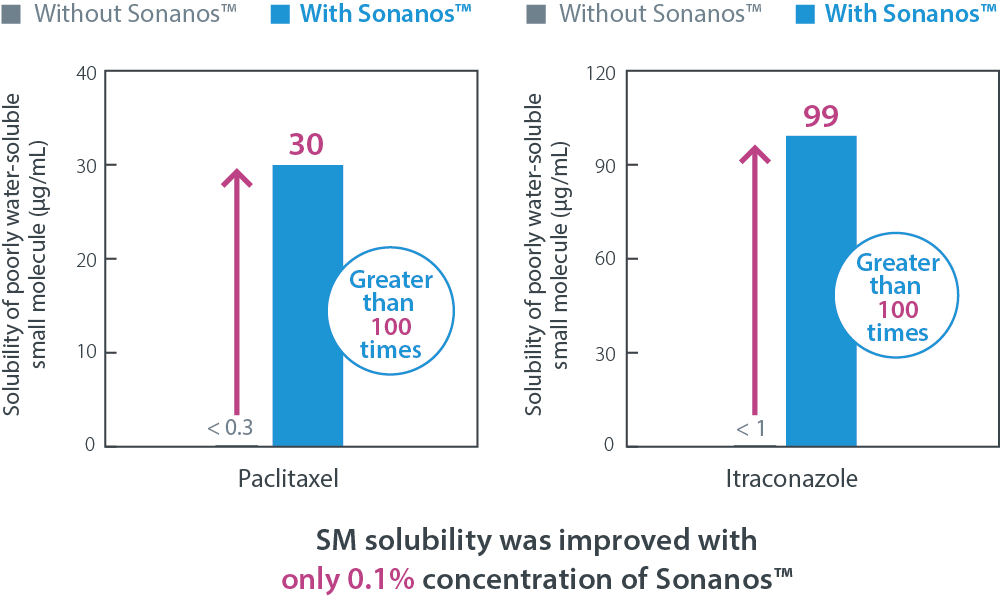
Solubility Enhancement of Small Molecule APIs
Sonanos™ DS can also function as an effective solubilizer for poorly water‑soluble small molecule APIs.
To evaluate its solubilization capacity,
poorly water‑soluble APIs were mixed with a Sonanos™ DS solution and subsequently filtered.
As shown below,
Sonanos™ DS (0.1%) demonstrated a clear solubility‑enhancing effect for paclitaxel and itraconazole,
which were used as model compounds.
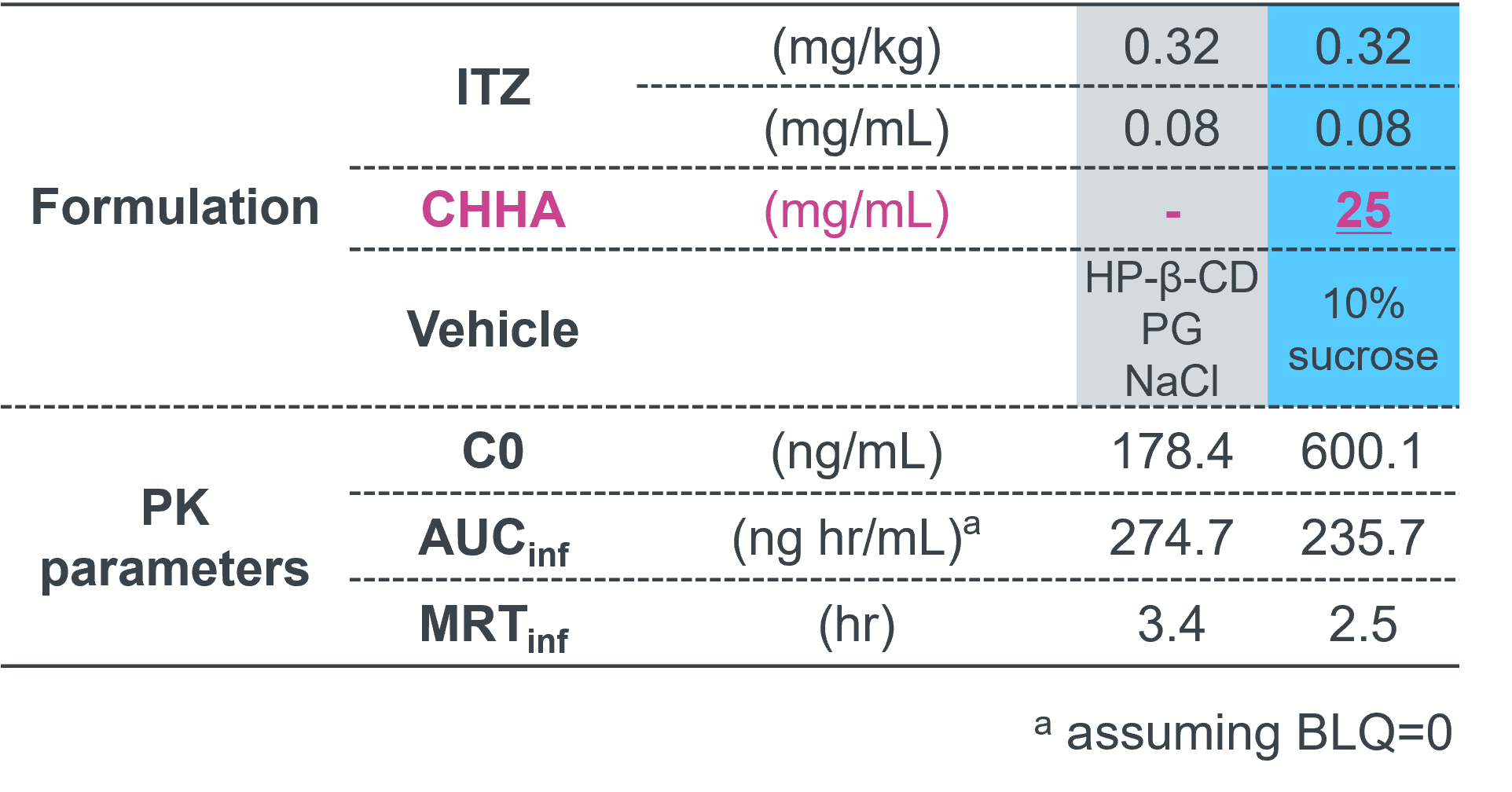
Effect of Sonanos™ DS on PK Profile of Small Molecule APIs
The impact of Sonanos™ DS on the PK profile of a small‑molecule API was further evaluated using itraconazole (ITZ)
as a model drug.
ITZ was selected because its half‑life (~4 hours) is substantially shorter than that of Sonanos™ nanogel particles
(~15 hours).
After intravenous administration of the ITZ formulation solubilized
with Sonanos™ DS to Sprague–Dawley rats (SD rats),
no significant changes in the PK profile were observed.
This result further supports the suitability of Sonanos™ DS
as a solubilizer and suggests rapid release of the encapsulated API following systemic administration.
- Animals: SD rats, male, 6-7 weeks
- IV, N = 4, 4.0 mL/kg
- Concentration measurements: HPLC